Abstract
Background
Gene therapy for hemophilia A has the potential to reduce the treatment burden for patients and their care providers by eliminating the need for regular factor VIII (FVIII) prophylaxis through long-term expression of endogenous FVIII at levels sufficient to provide bleed protection. Ongoing phase 3 gene therapy trials for hemophilia A show promise but can result in unpredictable FVIII expression of uncertain durability. Gene therapy must evolve to meet patient expectations of a durable, efficacious and safe treatment.
BAY 2599023 (AAVhu37.hFVIIIco) is the first, clinical stage adeno-associated virus (AAV) gene therapy vector, based on the AAVhu37 serotype. BAY 2599023 is a non-replicating AAV vector and contains a single-stranded DNA genome encoding a B-domain-deleted FVIII, under the control of a liver-specific promoter/enhancer combination, optimized for transgenic expression. The AAVhu37 capsid is a member of the hepatotropic clade E family and was selected based on preclinical studies demonstrating efficient, liver-directed FVIII gene transfer, favorable biodistribution and durable FVIII expression. Here, we report safety and FVIII activity levels achieved to date in this first-in-human, dose-finding study of BAY 2599023.
Methods
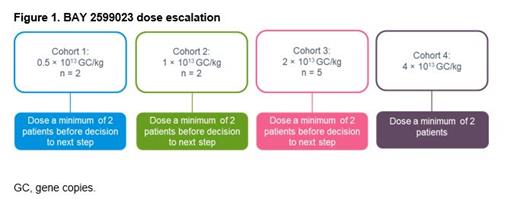
The ongoing BAY 2599023 phase 1/2, open-label, dose-finding study (NCT03588299) included male patients aged ≥18 years with severe hemophilia A, no history of FVIII inhibitors, no detectable neutralizing immunity against AAVhu37 (neutralizing antibody titer ≤5), and ≥150 exposure days to FVIII products. Patients received a single intravenous infusion of BAY 2599023 and were enrolled sequentially into three dose cohorts (0.5 × 10 13 GC/kg, 1.0 × 10 13 GC/kg and 2.0 × 10 13 GC/kg), each comprising at least two patients. Patients to be enrolled in a fourth cohort will receive a single infusion of 4 × 10 13 GC/kg (Figure 1). Primary endpoints were adverse events (AEs), serious AEs (SAEs) and AEs/SAEs of special interest (S/AESIs). The secondary endpoint was FVIII activity over time. Informed patient consent and ethics committee approval were obtained.
Results
Three cohorts of ≥2 patients each (N = 9) were enrolled sequentially (Figure 1). At the data cutoff (May 2021), FVIII activity data were available for the first eight patients.
BAY 2599023 delivered sustained FVIII expression levels for up to >23 months, with evidence of bleed protection. Patients in Cohorts 2 and 3 have all been off prophylaxis with FVIII products since approximately 6-12 weeks after gene transfer. To date, it has been observed that no spontaneous bleeds requiring treatment have been reported once FVIII levels >11 IU/dL were achieved. Of the 9 patients treated, 5 patients developed an AESI: mild/moderate alanine aminotransferase (ALT) elevations observed in Cohort 2 (n =1) and Cohort 3 (n = 3) were managed with corticosteroid treatment; another ALT elevation was reported as study-drug-related SAE in Cohort 3 (n = 1) but returned to normal a few weeks after interruption of the H2 blocker famotidine. The latest follow-up data for up to 28 months will be presented.
Conclusions
BAY 2599023 was designed to enhance efficacy and durability of FVIII expression with a favorable safety profile. Sustained FVIII levels allowed suspension of FVIII prophylaxis in the majority of patients, with asymptomatic ALT elevations that responded
to corticosteroids, making BAY 2599023 a key candidate in the evolution of gene therapy in hemophilia A.
Pipe: Genventiv: Consultancy; Regeneron/ Intellia: Consultancy; uniQure: Consultancy, Other; Spark Therapeutics: Consultancy; Takeda: Consultancy; Sanofi: Consultancy, Other; Sangamo Therapeutics: Consultancy; Roche/Genentech: Consultancy, Other; Pfizer: Consultancy; Novo Nordisk: Consultancy; Freeline: Consultancy, Other: Clinical trial investigator; HEMA Biologics: Consultancy; CSL Behring: Consultancy; Catalyst Biosciences: Consultancy; Biomarin: Consultancy, Other: Clinical trial investigator; Bayer: Consultancy; ASC Therapeutics: Consultancy; Apcintex: Consultancy; Grifols: Consultancy; Octapharma: Consultancy; Shire: Consultancy. Sheehan: BioMarin: Consultancy, Research Funding; Roche: Consultancy, Research Funding; Bayer: Consultancy, Research Funding. Coppens: Portola/Alexion: Research Funding; CSL Behring: Consultancy, Research Funding; Bayer: Consultancy, Research Funding; Roche: Research Funding; Daiichi Sankyo: Research Funding; Sanquin Blood Supply: Research Funding; uniQure: Research Funding; Medcon International: Consultancy; MEDtalks: Consultancy; Novo Nordisk: Consultancy; Pfizer: Consultancy; Sobi: Consultancy. Eichler: Takeda: Consultancy, Honoraria; BioMarin: Consultancy, Research Funding; Roche: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria; CSL Behring: Consultancy, Honoraria; Pfizer: Research Funding; Novo Nordisk: Consultancy, Research Funding; Biotest: Consultancy, Honoraria; Bayer: Consultancy, Research Funding. Linardi: Bayer: Current Employment. Wiegmann: Bayer: Current Employment. Hay: Pfizer: Consultancy, Research Funding; Inspiration: Consultancy, Honoraria; BioMarin: Consultancy, Honoraria; Roche: Consultancy, Research Funding; Bayer: Consultancy, Research Funding; Takeda: Consultancy, Research Funding; Spark Therapeutics: Consultancy, Research Funding. Lissitchkov: Bayer: Other: Principal Investigator of Clinical Trials; Catalist: Other: Principal Investigator of Clinical Trials; Grifols: Other: Principal Investigator of Clinical Trials.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal